With more than 10% of the world’s population obese and 40% overweight, obesity constitutes one of the most crucial health challenges. However, existing therapeutic options remain scarce and poorly efficient. Few years ago, scientists from the University of Geneva (UNIGE), Switzerland, discovered that the absorptive surface and function of the gut change due to certain external stimuli, such as exposure to cold. Today, by using different mouse models combined with human intestinal biopsies, they decipher the molecular mechanisms that govern this surprising organ plasticity, and uncover that increasing the food amount elevates the intestinal absorptive surface and function. Mechanistically, this is due to the enhanced expression of PPARα, a regulating protein necessary for the overeating-induced increase in the capacity of the gut to absorb calories. Furthermore, if high amounts of food increase the gut absorptive surface, food restriction can reverse the process and return it close to normal. This phenomenon could be reproduced using pharmacological and genetic approaches, thereby suggesting potential avenues to limit obesity. These results are published in the journal Nature Communications.
Throughout the world, hundreds of millions of adults and children are clinically obese; a condition closely associated with leading causes of death such as heart diseases or stroke. Obesity mainly results from an imbalance between energy expenditure and caloric uptake, which takes place in the gut where food, previously broken down, is absorbed and passes into the bloodstream for distribution throughout the body. To absorb enough calories, the intestinal wall is layered with millions of convolutions called villi and microvilli that together could cover the surface of a football field.
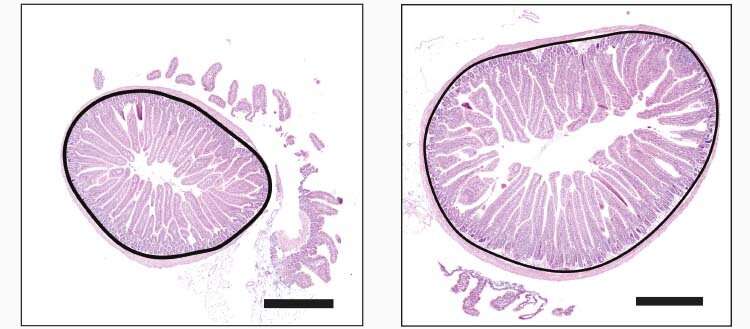
A striking and rapid effect
“A few years ago, we discovered that the gut could get longer or shorter according to environmental triggers and physiological needs”, recalls Mirko Trajkovski, professor in the Department of Cellular Physiology and Metabolism and the Diabetes Centre at UNIGE Faculty of Medicine, and corresponding author of this study. “We therefore wanted to understand what propels this remarkable intestinal plasticity.” Using different mouse models combined with human intestinal biopsies—3D artificial structures—the research team observed that the amount of food consumed was the main regulator of gut length. “We saw a relatively fast and physiologically striking response to elevating the amount of ingested food: gut length increased for over 30%, coupled with a major growth of the villi and microvilli, contributing to an enhanced caloric uptake capacity of the gut.” adds Mirko Trajkovski. Importantly, these changes were reversible: when the amount of food was reduced, the gut length and morphology were reverted close to normal.
A plasticity under the control of the PPARα protein
Gut expansion requires a lot of energy. The UNIGE scientists found that increasing the intestinal absorptive surface mobilizes different metabolic pathways in the gut, i.e. steps by which cells convert food into energy. While they found several potential pathways that may contribute to gut expansion, one, the PPARα pathway, was found to be indispensable. Indeed, PPARα is a protein that appears of critical importance for increasing the length of villi, as well as for increasing caloric uptake capacity from the food by raising the level of another protein, PLIN2, which, in turn, promotes formation of lipid droplets in the intestinal cells, thereby favoring fat absorption. By inactivating PPARα in the gut of mice, the researchers were able to confirm this mechanism. “Intestinal PPARα deletion, or its pharmacological inhibition, showed marked effects in reducing the absorptive function of the gut. The gut-specific PPARα inhibition was sufficient to revert the fat accumulation and obesity, as well as the glucose intolerance caused by high-caloric feeding”, explains Mirko Trajkovski.
Source: Read Full Article
